Peptide synthesis is a critical technology in modern biotechnology, pharmaceuticals, and life science research. As demand grows for high-purity custom peptides, understanding how peptide synthesis works—and choosing the right synthesis partner—has become increasingly important.
At D&L Peptides, we supply high-quality peptides and peptide synthesis solutions sourced to meet the needs of research, diagnostic, and commercial applications. This guide explains peptide synthesis in detail, including key methods such as solid-phase peptide synthesis (SPPS), essential reagents, and real-world applications.
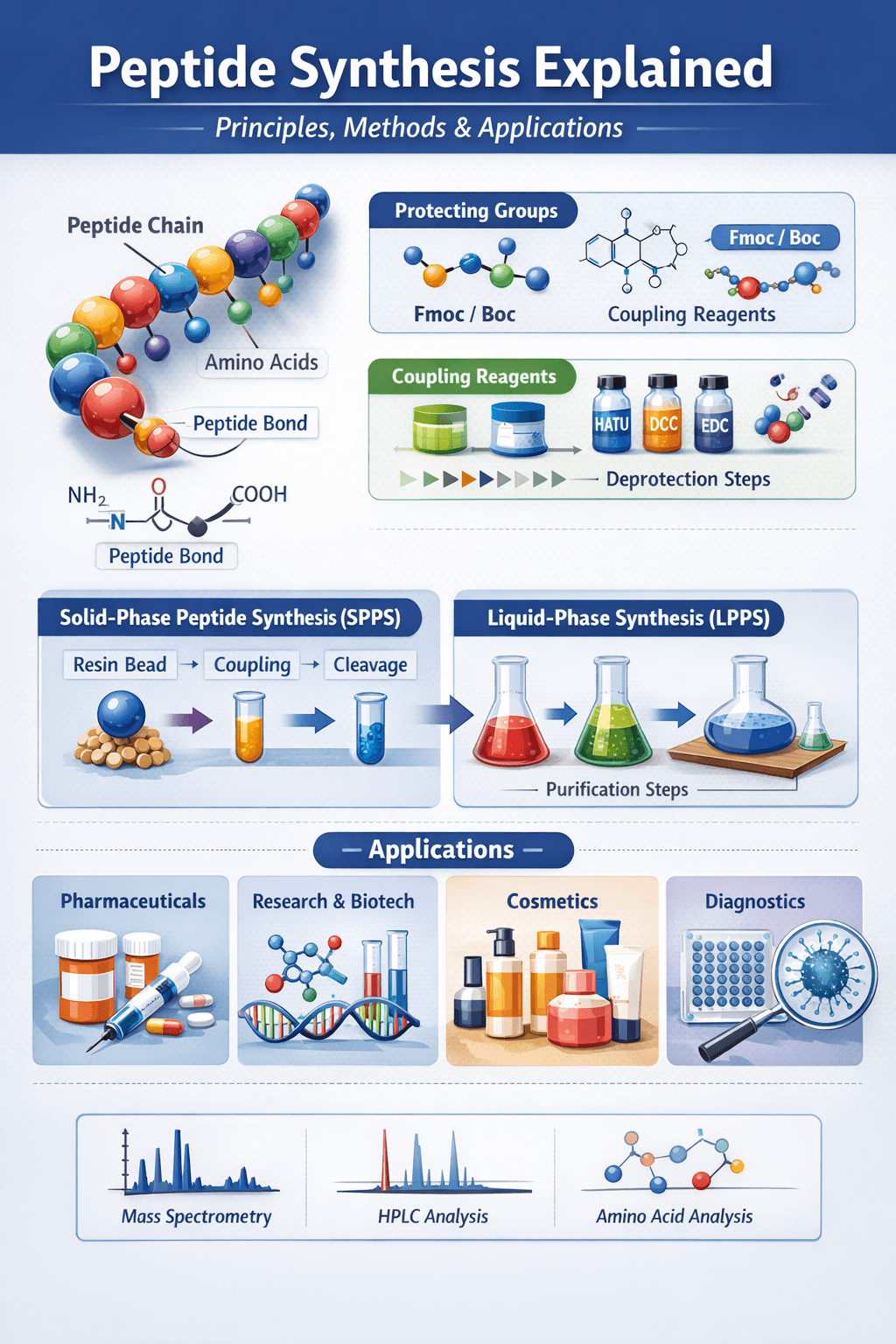
What Is Peptide Synthesis?
Peptide synthesis is the chemical process of constructing peptides by linking amino acids in a specific sequence. This controlled, stepwise method allows scientists to produce both naturally occurring peptides and custom-designed sequences.
Unlike protein biosynthesis in living cells, synthetic peptide production is carried out in vitro. This enables:
- Precise sequence control
- Custom modifications
- High purity and reproducibility
These advantages make synthetic peptides essential in pharmaceutical development, research, and diagnostics.
How Peptide Synthesis Works (Step-by-Step)
The peptide synthesis process follows a repeated cycle of chemical reactions:
- Attachment – The first amino acid is anchored to a solid support (resin)
- Deprotection – The protecting group is removed to expose the reactive site
- Coupling – The next amino acid is added using coupling reagents
- Repetition – Steps are repeated until the full sequence is assembled
- Cleavage and Purification – The peptide is released and purified (typically via HPLC)
Each step must be carefully controlled to ensure high yield and sequence accuracy.
Key Components in Peptide Synthesis
Protecting Groups (Fmoc and Boc)
Protecting groups prevent unwanted side reactions during synthesis.
- Fmoc (Fluorenylmethyloxycarbonyl) – widely used, base-labile
- Boc (tert-Butyloxycarbonyl) – acid-labile, used in specific workflows
These groups are removed at defined stages to allow controlled peptide elongation.
Coupling Reagents
Efficient peptide bond formation depends on high-performance coupling reagents, including:
- HATU
- DCC
- EDC
These reagents activate amino acids and improve reaction efficiency while minimising by-products.
Main Peptide Synthesis Methods
Solid-Phase Peptide Synthesis (SPPS)
Solid-phase peptide synthesis (SPPS) is the most widely used method in modern peptide production.
In SPPS, the peptide is built on a solid resin, allowing excess reagents to be removed easily. This simplifies the process and enables automation.
Benefits of SPPS:
- High efficiency and reproducibility
- Scalable production
- Reduced purification steps
- Ideal for custom peptide synthesis
D&L Peptides sources and supplies high-quality peptides produced using optimised SPPS systems, supporting a wide range of applications.
Liquid-Phase Peptide Synthesis (LPPS)
Liquid-phase peptide synthesis is performed entirely in solution. While it offers precise control, it requires purification after each step, making it more time-consuming.
This method is typically used in specialised or large-scale production scenarios.
Native Chemical Ligation (NCL)
For longer peptides and small proteins, native chemical ligation enables the joining of peptide fragments. This method is essential for synthesising complex or extended sequences beyond standard SPPS limits.
Challenges in Peptide Synthesis
Despite advances in technology, peptide synthesis presents several challenges:
- Aggregation of peptide chains, especially in hydrophobic sequences
- Incomplete coupling reactions, leading to impurities
- Reduced yield with longer peptides
At D&L Peptides, these challenges are addressed by sourcing peptides produced using optimised protocols, high-quality reagents, and rigorous process control.
Purification and Quality Control
High purity is essential for reliable results in research and commercial applications.
After synthesis, peptides are purified using high-performance liquid chromatography (HPLC). Analytical testing ensures quality and accuracy:
- Mass spectrometry (MS) – confirms molecular weight
- HPLC analysis – verifies purity
- Additional testing – as required for specific applications
DL Peptides maintains strict quality control standards to ensure consistent product performance.
Applications of Peptide Synthesis
Pharmaceuticals and Drug Development
Peptides are widely used as therapeutic agents due to their specificity and safety profile. They are applied in:
- Diabetes treatments (e.g., insulin analogues)
- Cancer therapeutics
- Metabolic and endocrine disorders
Research and Biotechnology
Custom peptides are essential tools in:
- Protein interaction studies
- Antibody production
- Epitope mapping
Diagnostics
Peptides are used in diagnostic assays, including ELISA and biomarker detection, where specificity is critical.
Cosmetics
Bioactive peptides are increasingly used in skincare formulations for anti-ageing and skin regeneration.
Recent Advances in Peptide Synthesis
Modern peptide synthesis continues to evolve with improved efficiency and sustainability:
- Automated peptide synthesis systems
- Microwave-assisted synthesis for faster reactions
- Greener chemistry approaches
- Advanced peptide modifications (e.g., cyclisation, PEGylation)
These innovations enable the production of more complex and stable peptides.
