Introduction
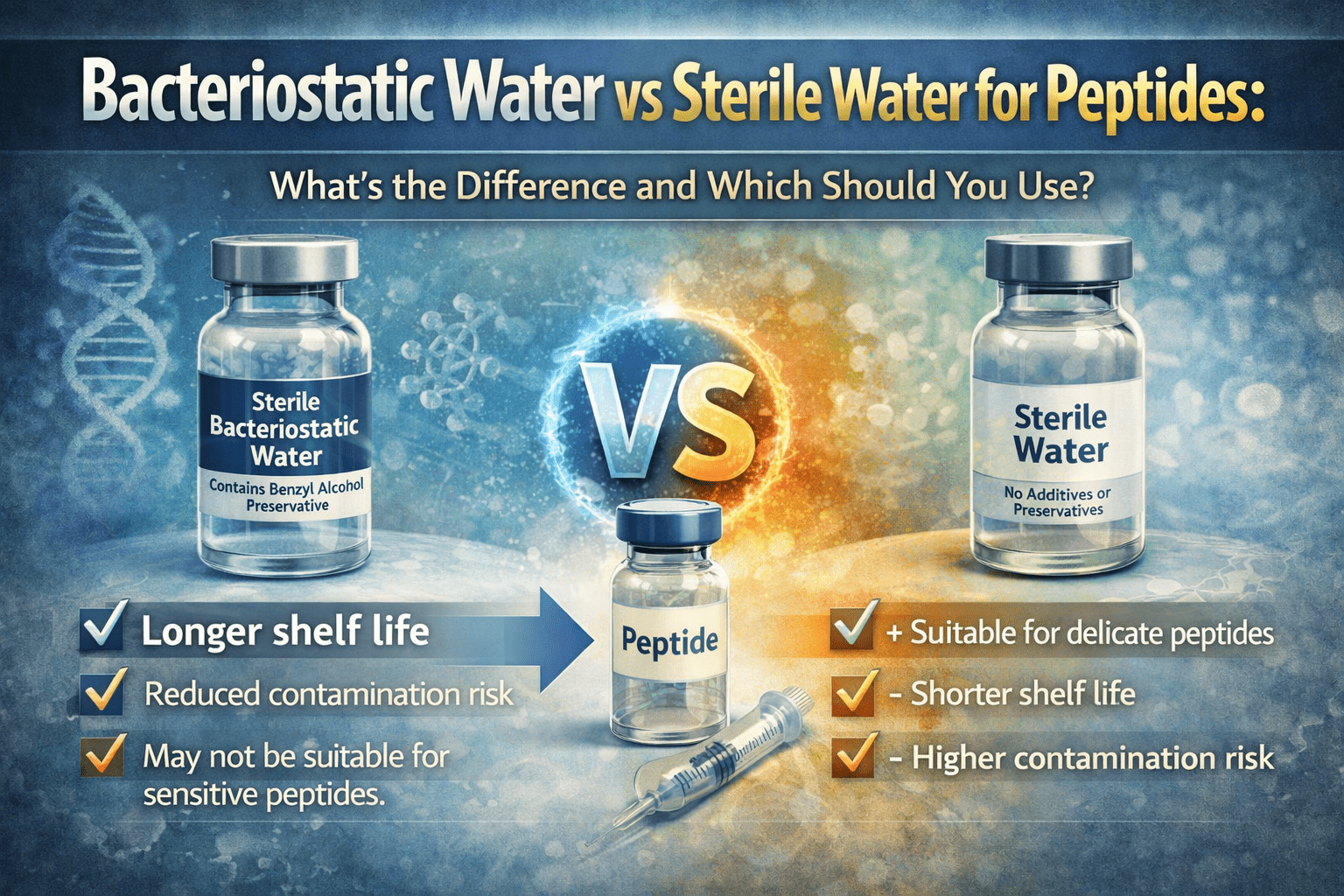
When reconstituting peptides, one of the most common decisions researchers face is whether to use bacteriostatic water or sterile water.
At first glance, the difference seems minor. Both are used to dissolve lyophilised peptides, and both are widely accepted in research settings. However, the choice can influence how a peptide behaves after reconstitution—particularly in terms of contamination risk and usability over time.
Understanding the distinction is important, especially when working with sensitive compounds or multi-use protocols.
What Is Sterile Water?
Sterile water is purified water that has been filtered and sterilised to remove microorganisms and contaminants.
It contains no additives or preservatives, making it a simple and clean solvent for peptide reconstitution.
Because of this, sterile water is often used in:
- Single-use preparations
- Controlled experimental setups
- Situations where additives are not desired
However, once a vial is accessed, sterile water provides no protection against microbial growth.
What Is Bacteriostatic Water?
Bacteriostatic water is sterile water that contains a small amount of preservative, typically benzyl alcohol. This additive helps inhibit the growth of bacteria after the vial has been opened.
This makes bacteriostatic water more suitable for:
- Multi-use peptide vials
- Repeated access over time
- Research setups requiring extended usability
It’s important to note that bacteriostatic water does not sterilise contaminated solutions—it simply slows bacterial growth.
The Key Difference
The core difference between bacteriostatic water and sterile water comes down to preservatives.
Sterile water contains none. Bacteriostatic water includes an agent designed to reduce microbial growth after reconstitution.
This difference becomes relevant once a peptide is mixed and stored, particularly if the vial will be accessed multiple times.
How This Affects Peptide Stability
A common misconception is that bacteriostatic water improves peptide stability. In reality, it does not prevent chemical degradation.
Once a peptide is reconstituted, it is subject to:
- Hydrolysis
- Oxidation
- Structural changes over time
These processes occur regardless of the solvent used.
The role of bacteriostatic water is not to preserve the peptide itself, but to reduce the risk of bacterial contamination during use.
👉 For a deeper explanation, see How Long Do Reconstituted Peptides Last
When to Use Sterile Water
Sterile water is typically preferred in controlled, single-use scenarios.
If a peptide is reconstituted and used immediately, the lack of preservatives is not an issue. In fact, some researchers prefer sterile water specifically because it introduces no additional compounds into the solution.
This approach is often used in tightly controlled experiments where consistency and simplicity are prioritised.
When to Use Bacteriostatic Water
Bacteriostatic water is more practical when a peptide will be used across multiple sessions.
If a vial is accessed repeatedly, the risk of introducing contaminants increases. The preservative in bacteriostatic water helps reduce this risk, making it a more suitable choice in these cases.
However, even with bacteriostatic water, peptides should not be stored indefinitely after reconstitution.
Common Misunderstandings
One of the most common misconceptions is that bacteriostatic water “extends peptide life.” While it helps manage contamination, it does not stop degradation.
Another misunderstanding is that sterile water is always inferior. In reality, it is simply suited to a different type of use—primarily single-use applications.
Choosing the wrong solvent won’t necessarily ruin a peptide, but misunderstanding their roles can lead to incorrect assumptions about stability.
Practical Considerations for Researchers
The decision between bacteriostatic and sterile water should be based on how the peptide will be used.
If the solution is intended for immediate use, sterile water is often sufficient. If the peptide will be accessed multiple times, bacteriostatic water provides an added layer of protection against contamination.
In both cases, proper storage and handling remain essential. Solvent choice is only one part of maintaining peptide integrity.
👉 See also:
🔬 Why Peptide Quality Still Matters
Even with correct solvent choice, results depend heavily on the quality of the peptide itself.
High-purity, properly lyophilised peptides:
- Reconstitute more cleanly
- Maintain structure more effectively
- Produce more consistent outcomes
At DL Peptides, all compounds are supplied in lyophilised form and batch-tested for purity and consistency.
👉 Explore Research-Grade Peptides
If you’re working with peptides that require precise handling, sourcing and preparation both matter.
- 👉 View Tesamorelin (Research Use Only)
- 👉 Explore CJC-1295, Ipamorelin & IGF-1
- 👉 Browse the full D&L Peptides catalogue
Final Thoughts
Bacteriostatic water and sterile water serve different purposes in peptide research.
The key distinction is not stability, but contamination control. Understanding this difference helps ensure that peptides are handled appropriately for the intended use.
In most cases, the right choice comes down to a simple question: will the peptide be used once, or multiple times?
Answer that correctly, and the rest becomes much clearer.
Frequently Asked Questions
Is bacteriostatic water better than sterile water for peptides?
Not necessarily. Bacteriostatic water helps reduce microbial growth, while sterile water contains no additives. The best choice depends on how the peptide will be used.
Does bacteriostatic water extend peptide shelf life?
No. It reduces contamination risk but does not prevent chemical degradation of the peptide.
Can sterile water be used for peptides?
Yes. Sterile water is commonly used for single-use reconstitution where contamination risk is minimal.
Why do researchers use bacteriostatic water?
It allows for safer multi-use of reconstituted peptides by inhibiting bacterial growth after vial access.
Do all peptides require bacteriostatic water?
No. The choice depends on the research setup, not the peptide itself.
References
Wang, W. (1999). Instability, stabilization, and formulation of liquid protein pharmaceuticals. International Journal of Pharmaceutics.
Manning, M. C. et al. (2010). Stability of protein pharmaceuticals. Pharmaceutical Research.
General peptide formulation and handling literature
